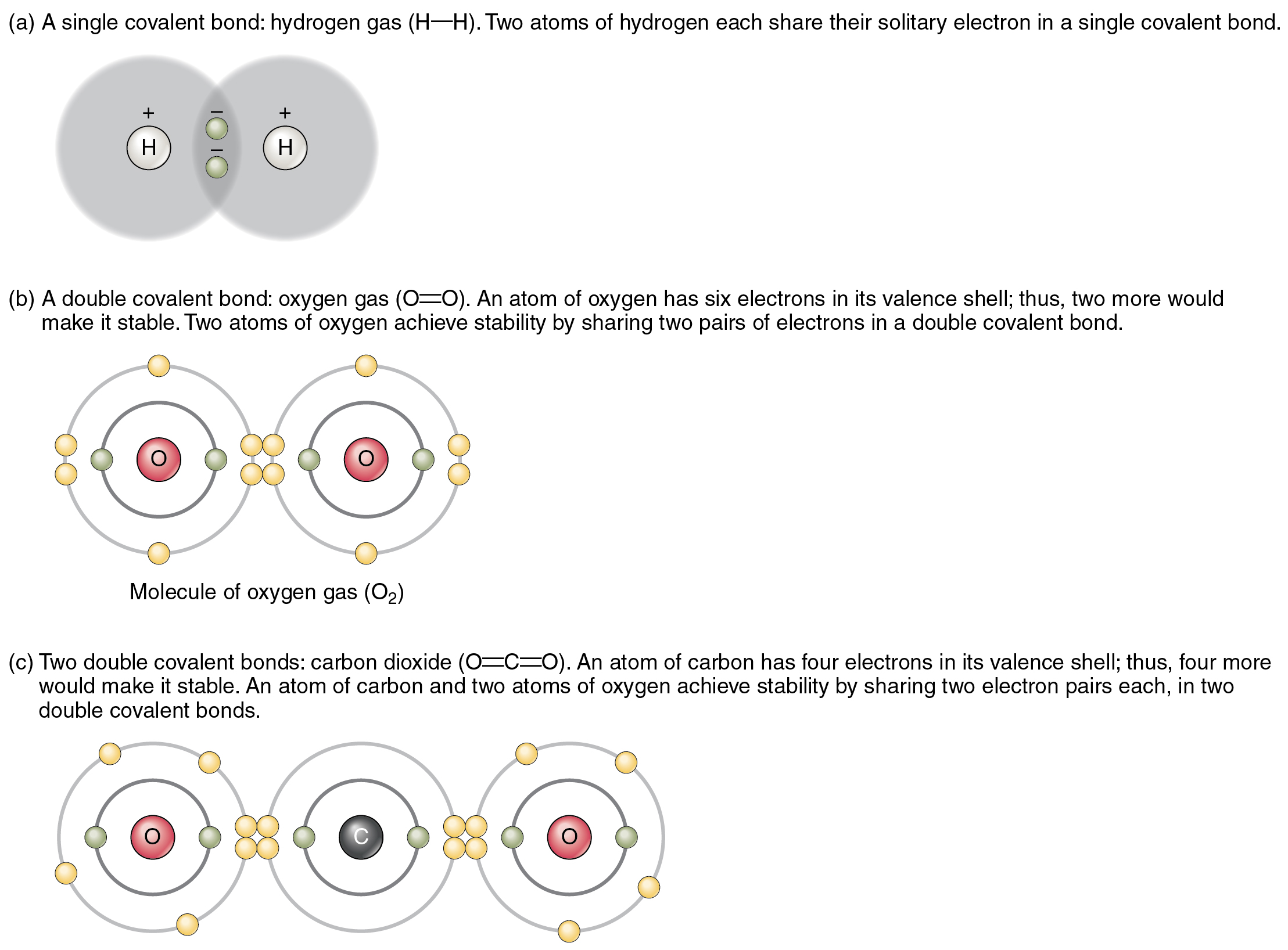
Can Carbon Form Ionic Bonds - The textbook also mentions that a.
Can Carbon Form Ionic Bonds - Web moreover, of all the elements in the second row, carbon has the maximum number of outer shell electrons (four) capable of forming covalent bonds. In general bonds of carbon with other elements are covalent bonds. Web ions and ionic bonds. In most cases, carbon shares electrons with other atoms. Figure 2.29 some elements exhibit a regular pattern of ionic charge.
Carbon does not form ionic bonds because it has 4 valence electrons, half of an octet. Carbon does not form ionic bonds because it has 4 valence electrons, half of an octet. Ad uslegalforms.com has been visited by 100k+ users in the past month Ionic bonds result from the attraction. Web organic carbon compounds are far more numerous than inorganic carbon compounds. Web when bonds are formed, energy is released and the system becomes more stable. To form ionic bonds, carbon molecules must either gain or.
Ionic Bond Definition, Types, Properties & Examples
Web aluminum and carbon react to form an ionic compound. Web ions and ionic bonds. Web we would like to show you a description here but the site won’t allow us. This is because carbon has 4 valence. One type of chemical bond is an ionic bond. Carbon does not form ionic bonds because it.
Chapter 5.6 Properties of Polar Covalent Bonds Chemistry LibreTexts
Web ions and ionic bonds. The most common type of bond formed by carbon is a covalent bond. Web aluminum and carbon react to form an ionic compound. Carbon does not form ionic bonds because it has 4 valence electrons, half of an octet. Figure 2.29 some elements exhibit a regular pattern of ionic charge..
Ionic Bonding Presentation Chemistry
Atoms interact with each other through the formation of chemical bonds. So what's different for carbon? If it is with another atom, a polar covalent bond is formed. Predict which forms an anion, which forms a cation, and the charges of each ion. Carbon does not form ionic bonds because it has 4 valence electrons,.
Chemical Bonds · Anatomy and Physiology
Web ions and ionic bonds. This is because carbon has 4 valence. To form ionic bonds, carbon molecules must either gain or. The most common type of bond formed by carbon is a covalent bond. Carbon most often forms a covalent bond with other atoms. Web organic carbon compounds are far more numerous than inorganic.
Ppt Atomic Structure And Chemical Bonding Powerpoint Presentation 7F5
Ionic bonds result from the attraction. In general bonds of carbon with other elements are covalent bonds. Web formation of ionic bonds involve removing electrons and there seems to be enough energy there. Web we would like to show you a description here but the site won’t allow us. When they do so, atoms form.
Carbon to Carbon Single, Double & Triple Bonds Surfguppy
The most common type of bond formed by carbon is a covalent bond. Carbon does not form ionic bonds because it has 4 valence electrons, half of an octet. So what's different for carbon? Web these four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in ccl 4 (carbon.
PPT Unit 1 Biochemistry The Chemistry of Life PowerPoint
In most cases, carbon shares electrons with other atoms. Web there are ionic carbides ($\ce{al4c3}$, $\ce{cac2}$, etc.), graphite intercalation compounds like $\ce{kc8}$, ionic derivatives of fullerenes and more. Web for example, copper can form ions with a 1+ or 2+ charge, and iron can form ions with a 2+ or 3+ charge. Web ions and.
Carbon Oxygen Triple Bond DaytonknoeMoses
Write the symbol for each. Less commonly, carbon forms ionic bonds. So what's different for carbon? In most cases, carbon shares electrons with other atoms. Web aluminum and carbon react to form an ionic compound. In general bonds of carbon with other elements are covalent bonds. Carbon does not form ionic bonds because it has.
Four covalent bonds. Carbon has four valence electrons and here a
One type of chemical bond is an ionic bond. Web moreover, of all the elements in the second row, carbon has the maximum number of outer shell electrons (four) capable of forming covalent bonds. To form ionic bonds, carbon molecules must either gain or lose 4 electrons. So what's different for carbon? Carbon most often.
The 4 Types of Bonds Carbon Can Form Video & Lesson Transcript
In general bonds of carbon with other elements are covalent bonds. Write the symbol for each. Predict which forms an anion, which forms a cation, and the charges of each ion. Web for example, copper can form ions with a 1+ or 2+ charge, and iron can form ions with a 2+ or 3+ charge..
Can Carbon Form Ionic Bonds Web formation of ionic bonds involve removing electrons and there seems to be enough energy there. So what's different for carbon? This is because carbon has 4 valence. Carbon does not form ionic bonds because it has 4 valence electrons, half of an octet. Write the symbol for each.
Ad Uslegalforms.com Has Been Visited By 100K+ Users In The Past Month
Atoms interact with each other through the formation of chemical bonds. In general bonds of carbon with other elements are covalent bonds. Web formation of ionic bonds involve removing electrons and there seems to be enough energy there. Figure 2.29 some elements exhibit a regular pattern of ionic charge.
If It Is With Another Atom, A Polar Covalent Bond Is Formed.
Carbon does not form ionic bonds because it has 4 valence electrons, half of an octet. Web moreover, of all the elements in the second row, carbon has the maximum number of outer shell electrons (four) capable of forming covalent bonds. Web organic carbon compounds are far more numerous than inorganic carbon compounds. Web there are ionic carbides ($\ce{al4c3}$, $\ce{cac2}$, etc.), graphite intercalation compounds like $\ce{kc8}$, ionic derivatives of fullerenes and more.
Carbon Does Not Form Ionic Bonds Because It Has 4 Valence Electrons, Half Of An Octet.
Ionic bonds result from the attraction. One type of chemical bond is an ionic bond. Write the symbol for each. Ad over 27,000 video lessons and other resources, you're guaranteed to find what you need.
The Textbook Also Mentions That A.
To form ionic bonds, carbon molecules must either gain or lose 4 electrons. Web we would like to show you a description here but the site won’t allow us. Web these four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in ccl 4 (carbon tetrachloride) and silicon in sih 4 (silane). Web ions and ionic bonds.



.PNG)